Instead, it tries to depict stereochemical relationships: whether two substituents are on the same face of the ring or opposite faces of the ring. A Haworth projection does not try to do that. The chair drawing or "diamond lattice projection" shows these relationships pretty well. Also, substituents on the atoms in the ring can be found above the ring, below the ring, or sticking out around the edge of the ring. The chair drawing shows that relationship, but in a Haworth projection, the ring is drawn as though it were flat. For example, in a six-membered ring, the atoms in the ring adopt a zig-zag, up-and-down pattern in order to optimize bond angles. Haworth projections don't reflect the real shape of the ring. The most common way of drawing these rings are in "Haworth projections". Five-membered rings are called "furanoses" and six-membered rings are called "pyranoses". As a result, five- and six-membered rings are very common in sugars. The hemiacetal forms when a hydroxyl group along the carbon chain reaches back and bonds to the electrophilic carbonyl carbon. That's because the carbonyl is usually "masked" as a hemiacetal. In fact, if you have seen drawings of sugars before, you might not have noticed the carbonyl. Can a sugar react with itself? Of course. In just one molecule, we have both an electrophile (the carbonyl) and a number of nucleophiles (the hydroxyls). This sort of structure presents lots of possibilities for reactions. In general, a sugar is a molecule that contains an aldehyde or a ketone, and every carbon other than the carbonyl carbon has a hydroxyl group attached to it. One strict definition of a carbohydrate is a polyhydroxylated aldehyde or ketone. The term "sugar" is often applied to any simple carbohydrate. They carry specialized chemical reagents into enzymes where reactions essential to life are carried out. They aid in homebody security and defense operations by forming molecular codes on the surfaces of cells that identify whether the cell is one of our own or an intruder. For example, they lend structural support in the backbone of DNA. Although their best-known role is in energy storage in the form of glucose and starch, carbohydrates play a number of other roles. Notice how only the direction of the hydroxyl group on C 1 is different.\)Ĭarbohydrates are an important class of biological molecules. The paper and molecular structures of the product are shown in a similar orientation, with C 1 toward the right. 
Then click on the Close Ring button to see which anomer is formed from that configuration. For example, α glucose rotates polarized 589 nm light more than β glucose.Ĭlick on the image to switch the configuration of C 1. 
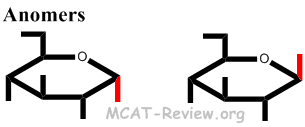
If they are on the same side, C 1 is said to be the β anomer.ĭo the α and β anomers have the same properties?īecause they have the same formula, glucose's α and β anomers share many properties, such as their solubility in water, heat of combustion, and ability to reduce certain compounds. If the hydroxyl group on C 1 and the -CH 2OH group on C 5 are on opposite sides of the six-membered ring, C 1 is said to be the α anomer. These two forms are identical except for the configuration around C 1. Depending on the orientation of C 1 when the C 5 hydroxyl bonds to it, two different forms can result.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
